

The future depends on delivering affordable healthcare to the developing world such as low-cost, portable pre-screening ophthalmology
Of the 285 million visually impaired across the globe, 40 million are completely blind.1 Forus Health is based in India, where 15 million people are blind.2 This is largely due to a lack of accessible health care, as the ophthalmologist-to-patient ratio is approximately 1:60,000. Many other countries face a similar lack of accessible eye care.
The leading causes of vision impairment are uncorrected refractive errors, cataracts, age-related macular degeneration, glaucoma, diabetic retinopathy, trachoma, and corneal opacity (Figure 1). When left untreated, these conditions worsen and may lead to complete blindness. People 50 years and older account for 82% of all blind, and this percentage continues to increase as life expectancy increases.1
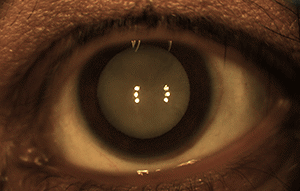
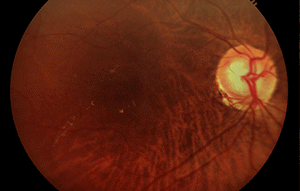
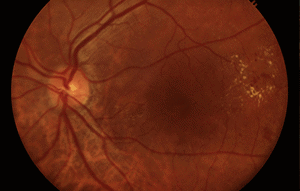

More than 15 million babies are born prematurely every year globally, which is more than 1 in every 10 births.3 60% of these premature infants are born in South Asia and Africa, and poorer families are at a higher risk. Low birth weight premature infants are at risk of retinopathy of prematurity (ROP), an eye disorder where fragile blood vessels grow abnormally, which scars their retina and can blind them by pulling it out of position. When detected and corrected early on, ROP can be treated and blindness can be avoided.
Forus Health’s mission is to eradicate preventable blindness by creating mass screening ecosystems supported by portable, affordable ophthalmic devices and digital health platforms.
Forus Health’s ophthalmic devices required a variety of optical components including lenses, mirrors, and polarizers. Early versions of their products yielded excellent images at the visible spectrum, yet poor images in the NIR. Bright reflections were evident at the center of the image and interfered with the diagnostic procedure. The visible spectrum allows for normal examination of the entire retina surface and cornea, but high quality NIR images are required for continuous observation without any irritation to the eyes.
Forus Health was also a startup company, which inherently involves demanding product deadlines and racing to secure investors. Startups are traditionally both time- and cost-sensitive, requiring them to find creative ways to develop their prototypes while minimizing expenses.
Edmund Optics was able to help Forus Health develop prototypes for their ophthalmic devices and support them as they found success and moved to production. Forus Health incorporated standard spherical optical components such as plano-convex and achromatic lenses in conjunction with specialized mirrors and filters. Edmund Optics adjusted the anti-reflection coatings of the components to cover longer wavelengths out to the NIR, which remedied the reflections and noise in the system during NIR imaging. R. J. Venkatramanan, Chief of Manufacturing and QA & RA at Forus Health, stated that "the Edmund technical team helped us to understand the product and improve the quality."

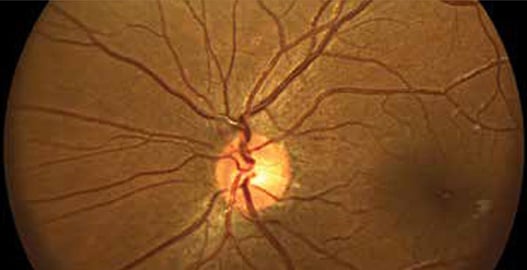
As Forus Health gained traction and scaled up their production, they came out with new models to address different challenges in combatting preventable blindness. The 3nethra classic, Forus Health’s flagship product, is a digital non-mydriatic fundus camera used to quickly capture and transmit images of the eye’s posterior and anterior surfaces. Mydriatic devices dilate the pupil before capturing images. The 3nethra neo differs from the classic in that it is mydriatic, ergonomically designed, lightweight, and hand-held for capturing 120° high-resolution images of the posterior and anterior segments of newborn eyes. The contact device is easily operated by qualified clinicians and can be deployed in versatile clinical environments such as hospitals, operating rooms, and Neonatal Intensive Care Units (NICU).

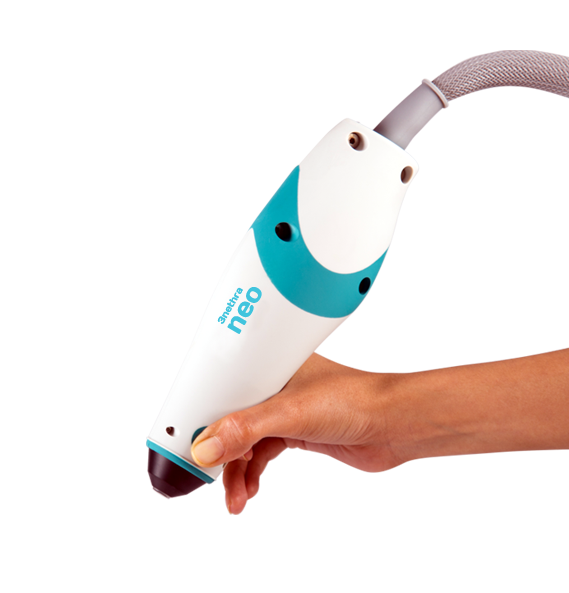
All versions of the 3nethra are compact enough to be easily transported and deployed in rural regions, allowing Forus Health to provide much needed eye care to countries including India, the Philippines, Mexico, Guatemala, Ethiopia, and Myanmar. Forus Health’s customers consist of a broad set of healthcare providers, such as ophthalmology hospitals and clinics, diabetic and diagnostic clinics, and vision centers. Forus Health continues to work towards creating a future where preventable blindness is a thing of the past.
or view regional numbers
QUOTE TOOL
enter stock numbers to begin
Copyright 2023, Edmund Optics Inc., 101 East Gloucester Pike, Barrington, NJ 08007-1380 USA
California Consumer Privacy Acts (CCPA): Do Not Sell or Share My Personal Information
California Transparency in Supply Chains Act
This content may include material that has been generated or modified using artificial intelligence (AI).
The FUTURE Depends On Optics®